 Introduction to control of documents & records – Use of LIMS for managing records.Implementation of a LIMS, in the context of ISO 17025ĭay One: Determination of Course Goals & Introduction to ISO 17025 Requirements.Basic guidelines on the design of a LIMS.Definition of Laboratory accreditation: Accreditation Bodies (AB’s) and Multilateral Agreements (MLA, MRA, ILAC) on cross-frontier recognition of accreditation.  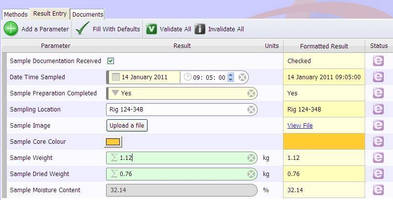
Technical requirements of ISO 17025 Personnel, Equipment, Traceability, Reference standards, Sampling, Quality assurance of results, Test Certificates, O & I’s.Management requirements of ISO 17025 Quality manual, Document control, Tenders, Suppliers, Service to the customer, Internal audits.Regulatory Authorities and organizations providing recognition). 
In addition, Management and Technical personnel of analytical laboratories will recognize the dire need of implementation of the Standard within their Organization in order to satisfy the needs of their customers and general market needs (e.g. The training course will also demonstrate the compliance of ISO 17025 with those ISO 9001 and GLP (Good Laboratory Practice) requirements that are relevant to the scope of testing services. In particular, the importance of LIMS implementation in meeting the traceability requirements of ISO 17025 will be addressed. This comprehensive training course will illustrate the ISO17025 requirements for testing laboratories, relevant to the operation of their management system, technical competency, the validity of analytical results, and the use of Laboratory Information Management Systems (LIMS) as a tool in satisfying the above. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
